Abdominal Aortic Aneurysms: Epidemiological, Clinical and Echographic Aspects in The Health Structures of Lomé and At The Kara University Hospital (TOGO)
Abstract
Objective
To describe the epidemiological, clinical and arterial Doppler ultrasound aspects of abdominal aortic aneurysm (AAA).
Material and Methods
This is a prospective study with descriptive and analytical aims carried out from January 2019 to March 2022 (3 years) at the cardiovascular exploration center (CEC) "Saint-Esprit" of the association of the people's doctors for the research on cardiovascular diseases (AMP-MCV), at the clinic "Le Printemps" and at the University Hospital of Kara. All patients who underwent arterial Doppler ultrasound of the lower limbs for any reason in these centers during the study period were included. Ultrasound scans were performed or confirmed by a physician with a diploma specializing in vascular Doppler ultrasound.
Results
We retained 32 Doppler ultrasound scans out of 739 performed for AAA with a frequency of 4.38%. The mean age was 73.5± 9.07 years. The sex ratio M/F was 1.4. The cardiovascular medical history (84.38%) found was: hypertension (33.33%), diabetes (18.52%), dyslipidemia (33.33%) and a combination of diabetes and hypertension (14.81%). The circumstances of discovery were: downstream MI ischemia (72%), incidental finding on abdominal ultrasound (15.63%) and De bakey sign (6.25%). The location of the aneurysms was subrenal (84.4%), suprarenal (9.4%) and both (6.2%). AAAs were fusiform (71.9%) and saccular (28.1%). The size of the aneurysms was greater than 5 cm in 78.17% of patients. High blood pressure increases the probability of the occurrence of subrenal AAA while dyslipidemia increases the occurrence of fusiform AAA. Diabetes increases the probability of occurrence of AAA larger than 7cm. AAAs were thrombosed in 75% of patients. There were extensions of the aneurysms to the iliac arteries in 70.59%. Downstream embolic complications represented 46.88%, in the form of occlusions and tight stenoses. Spontaneous mortality was 81.24%.
Conclusion
Arterial Doppler ultrasonography is a very important tool in the screening, positive and lesion diagnosis of AAA. The risk factors for AAA are: age over 60 years, male gender, hypertension, diabetes and dyslipidemia. Early detection remains essential, because the spontaneous prognosis is poor.
Author Contributions
Academic Editor: Sasho Stoleski, Institute of Occupational Health of R. Macedonia, WHO CC and Ga2len CC
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2023 Machihude PIO, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Aneurysm of the abdominal aorta (AAA) is a localized dilation of the abdominal aorta by at least half its normal diameter resulting in permanent loss of edge parallelism, turbulence of blood flow, and thrombus formation 1. These aneurysms can be saccular or fusiform 2. AAA is a serious condition that has a decisive impact on the vital prognosis 3. The prevalence is higher in men and increases with age. It is estimated between 2 and 6% of subjects over 65 years old in the USA 4.
In Africa, a study conducted on a population at risk of AAA in Benin in 2014 noted 1 case of AAA out of 245 patients selected, i.e. 0.41% of the population screened 5. As AAA is usually an asymptomatic pathology in 80% of cases, its discovery is most often fortuitous, during a paraclinical examination 6. Apart from inflammatory aneurysms, the occurrence of abdominal pain marks a turning point in the evolution of AAA. It is a sign of significant fissuring or compression and, above all, raises the fear of rupture leading to sudden death 7, 8. The overall mortality rate for patients with ruptured abdominal aortic aneurysms is 80 to 95% 7. Thus, in France, the French National Authority for Health (HAS) recommends targeted, opportunistic screening in patients with risk factors for atherosclerosis. Ultrasound allows the screening and diagnosis of AAAs 9 because of its non-invasive, inexpensive, non-irradiating nature and its hemodynamic advantages 10. In order to overcome the paucity of literature on AAAs in Togo and to determine the interest of early detection by arterial Doppler ultrasound of AAAs, we proposed to carry out this study, the main objective of which was to describe the epidemiological, clinical, and sonographic aspects of abdominal aortic aneurysms. The specific objectives were: to describe the sociodemographic characteristics of patients with AAA; to describe the circumstances of discovery of AAA in Togo; to describe the cardiovascular risk factors of AAA in Togo; to describe the Doppler ultrasound lesions of AAA.
Method
The cardiovascular exploration center (CEC) "Saint-Esprit" of the Association of People's Physicians for Research on Cardiovascular Diseases (AMP-MCV) in Lomé, the clinic "Le Printemps à Lomé" and the cardiology department of the CHU-Kara (located 450 km from Lomé) served as the setting for our study. There were no vascular exploration activities as such in the two cardiology departments of the two university hospitals of Lomé.
This was a prospective study with descriptive and analytical purposes that took place from January 2019 to March 2022 (3 years 2 months).
We included all patients who had undergone arterial Doppler ultrasound of the lower limbs, regardless of the reason, age, and sex in the centers selected for the study and during the study period.
AAA and its characteristics had to be confirmed on arterial Doppler ultrasound by the physician-cardiologist specialized in vascular Doppler explorations.
The performance of abdominal aortic angioscan was not essential for the inclusion of patients in this study.
Patients referred for suspected AAA in whom we did not have confirmation on arterial Doppler ultrasound were not́ retained in the study.
The same applies to patients with abdominal aortic dilatation less than 30 mm. Nonconsent to participate in the survey was also eliminatory.
In accordance with the technique for performing lower extremity arterial ultrasound, we always started and systematically explored the aorto-iliac axes in all patients.
AAA was defined as a rounded arterial vascular structure in cross section with a diameter greater than or equal to 30 millimeters that may have an anechogenic image corresponding to the aortic lumen and a more echogenic image corresponding to the thrombus. Longitudinal sections allowed us to identify the shapes of these AAAs, their location and extension. Extension to the visceral and iliac arteries was defined by an increase of one and a half times the normal size of these arteries. Arterial stenoses were defined as high peak systolic velocity with spectral dispersion and significant downstream pressure drop with flow demodulation. Occlusions were defined by 2-dimensional visualization of total or >90% obstruction (subocclusion) of the artery associated with an absence of pulsed or color Doppler at the artery or the presence of abutting flow.
Patients or accompanying persons were asked to answer questions on a predeveloped survey form.
Arterial Doppler ultrasounds were performed or confirmed by a cardiologist certified in vascular Doppler ultrasound.
Ultrasound checks were scheduled for 3 months with the possibility of a telephone link at any time.
For each patient, we were interested in sociodemographic data (age, sex); medical history, particularly cardiovascular and family AAA; and arterial Doppler ultrasound data.
All data were initially collected on a predefined survey form. Data were analyzed by Rstudio software version 4.1.0. We performed a descriptive and then univariate and multivariate analytical analysis. The qualitative variables were presented as numbers and frequencies, the quantitative variables as mean, standard deviation and extremes. The statistical tests used were the Pearson Chi-square test for qualitative variables and the Mann-Whitney test for quantitative variables. The significance level was set at 0.05. Univariate and multivariate logistic regression was performed to search for factors associated with aneurysm location, size, and shape.
Because ethical and deontological rules govern scientific research, we kept the patients anonymous in the evaluation forms to respect medical confidentiality.
Results
Sociodemographic Data
During the study period, out of 729 arterial Doppler ultrasounds performed, 32 (4.38%) were selected according to our inclusion criteria in the three selected centers in the country.
The age of the included patients ranged from 53 to 95 years, with a mean of 73.5± 9.07 years. The most represented age group was 66 to 75 years (43.75%). Males represented 18 cases (56.25%) with a sex ratio M/F of 1.4 (Table 1).
Table 1. Distribution of patients according to sex and age| Male | Female | |||
| n | (%) | n | % | |
| 50– 65 | 4 | 09,38 | 02 | 09,38 |
| 66 – 75 | 9 | 28,13 | 05 | 15,63 |
| 76 – 85 | 5 | 12,50 | 05 | 18,75 |
| 86 – 95 | 0 | 00,00 | 02 | 06,25 |
| Total | 18 | 56,25 | 14 | 43,75 |
Cardiovascular Medical History and Risk Factors for AAA
The patients included in this study had a cardiovascular medical history in 84.38% (27 patients). High blood pressure (28.13%), diabetes (15.63%), and dyslipidemia (28.13%) were the major cardiovascular risk factors present in our patients. There was no smoking or family aneurysm in our series.
Circumstances of Discovery
In 25 patients (78.13%), AAAs were discovered by downstream ischemia in the lower limbs, including 7 (28%) acute ischemia and 18 (72%) chronic ischemia. In 5 patients (15.63%) it was an incidental finding on abdominal ultrasound (Table 2).
Table 2. Distribution of patients according to the circumstances of discovery| Numbers (n) | Frequencies (%) | |
| Ischémie chroniqueChronic ischemia | 18 | 56,25 |
| Diabetic foot (gangrene) | 5 | 15,63 |
| AOMI stage III | 8 | 25,00 |
| AOMI stage IV (gangrene) | 5 | 15,63 |
| Acute MI ischemia | 7 | 21,88 |
| Incidental discovery | 5 | 15,63 |
| Flailing abdominal mass | 2 | 06,25 |
Arterial Doppler Ultrasound Data: Location of the Aneurysm
AAAs were subrenal in 27 patients (84.83%). Statistical analysis showed no significant association between age groups and aneurysm site. Similarly, there was no significant association between sex and AAA site.
High blood pressure was associated with the suprarenal location of the aneurysms with a p=0.012 and dyslipidemia with the subrenal location with a p=0.004 (Table 3).
Table 3. Distribution of patients according to cardiovascular medical history and aneurysm location| Subrenal | Sus renal | Sub and sus renal | |||||||
| n (N=27) | n/N | P | n (N=3) | n/N | P | n (N=2) | n/N | p | |
| HTA | 7 | 25,92 | 0,52 | 2 | 66,67 | 0,01 | 00 | 00 | 0,38 |
| Diabetes | 4 | 14,81 | 0,40 | 0 | 00,00 | 0,16 | 1 | 50 | 0,44 |
| HTA+Diabetes | 2 | 7,40 | 0,22 | 1 | 33,33 | 0,42 | 1 | 50 | 0,48 |
| Dyslipidemia | 9 | 33,33 | 0,004 | 0 | 00,00 | 0,13 | 0 | 00 | 0,18 |
Arterial Doppler Ultrasound Data: Shape of the Aneurysms
AAAs were fusiform in 23 patients (71.9%). The saccular aneurysm was present in 9 patients (28.1%). Figure 1 and (Figure 2).
Figure 1.Partially thrombosed 5.2cm fusiform suprarenal AAA with extension to the superior mesenteric artery in a 59-year-old subject (CEC 'Saint-Esprit' of the AMP-MCV).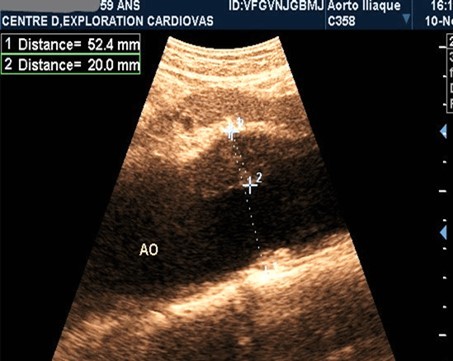
Figure 2.Partially thrombosed 5.2cm fusiform suprarenal AAA with extension to the superior mesenteric artery in a 59-year-old subject (CEC 'Saint-Esprit' of the AMP-MCV).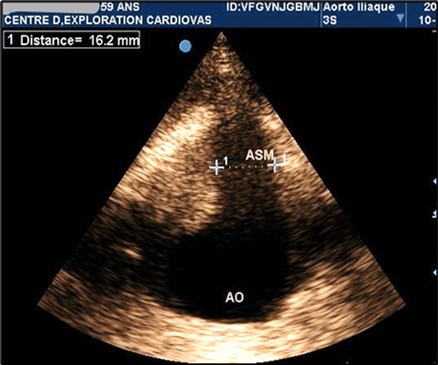
Statistical analysis did not show a significant association between age and the shape of the AAAs. Similarly, the statistical analysis did not show a significant association between age groups and aneurysm shape. High blood pressure was associated with the saccular shape of the aneurysms with a p=0.01 (Table 4).
Table 4. Distribution of patients according to cardiovascular medical history and aneurysm shape| Fusiform Aneurysm | Saccular Aneurysm | ||||
| n (N=23) | n/N | n (N=9) | n/N | p | |
| HTA | 6 | 26,08 | 3 | 33,33 | 0,01 |
| Diabetes | 4 | 17,39 | 1 | 11,11 | 0,23 |
| HTA+Diabetes | 2 | 8,70 | 2 | 22,22 | 0,17 |
| Dyslipidemia | 7 | 30,43 | 2 | 22,22 | 0,07 |
Arterial Doppler Ultrasound Data: Aneurysm Size
The mean size was 7.43 cm in this study. AAAs of size greater than or equal to 5 were found in 25 patients (78.14%) Table 5.
Table 5. Distribution of patients according to aneurysm size| Height in cm | Numbers (n) | Frequencies (%) |
|---|---|---|
| (3-4) | 01 | 03,13 |
| (4-5) | 06 | 18,75 |
| (5-6) | 07 | 21,88 |
| (6-7) | 07 | 21,88 |
| ≥7 | 11 | 34,38 |
| Total | 32 | 100,0 |
Statistical analysis showed no significant association between sex and aneurysm size. Patients with diabetes had larger AAA sizes p= 0.008 (Table 6).
Table 6. Distribution of patients according to cardiovascular medical history and aneurysm size| CRF | (3-4) | (4-5) | (5-6) | (6-7) | ≥7 | P | |||||
| n(N=1) | n/N | n(N=5) | n/N | n(N=6) | n/N | n(N=7) | n/N | n(N=11) | n/N | ||
| HTA | 1 | 100,00 | 2 | 40,00 | 4 | 66,67 | 2 | 28,57 | 0 | 00,00 | 0,216 |
| Diabetes | 0 | 0,00 | 0 | 00,00 | 0 | 00,00 | 0 | 00,00 | 5 | 45,45 | 0,008 |
| HTA+Diabetes | 0 | 0,00 | 2 | 40,00 | 2 | 33,33 | 0 | 00,00 | 0 | 00,00 | 0,21 |
| Dyslipidemia | 0 | 0,00 | 1 | 20,00 | 0 | 00,00 | 5 | 71,42 | 3 | 27,27 | 0,52 |
Arterial Doppler Ultrasound Data: Thrombotic Complications and Downstream Lesions in the Lower Limbs
Aneurysms were thrombosed in 24 patients (75%). (Figure 3) and (Figure 4) Arterial occlusion was found in 11 patients (73.33%) and significant stenosis in 4 patients (36.36%). (Table 7).
Table 7. Distribution of patients according to the location of occlusions and stenosis| Numbers (n) | Frequencies (%) | |
| Occlusion (N=11) | 11 | 34,38 |
| Iliac arteries | 2 | 18,18 |
| Femoral arteries | 5 | 45,45 |
| Popliteal arteries | 1 | 09,09 |
| Tibial arteries | 2 | 18,18 |
| Fibular arteries | 1 | 09,09 |
| Stenosis (N=4) | 4 | 12,50 |
| Iliac arteries | 1 | 25,00 |
| Femoral arteries | 1 | 25,00 |
| Popliteal arteries | 0 | 00,00 |
| Tibial arteries | 2 | 50,00 |
| Fibular arteries | 0 | 00,00 |
Figure 3.Large fusiform subrenal abdominal aortic aneurysm of 9.28cm thrombosed at 4/5 in an 82-year-old woman (CEC ‘Saint-Esprit’ from AMP-MCV).
Figure 4.Large fusiform subrenal abdominal aortic aneurysm of 9.28cm thrombosed at 4/5 in an 82-year-old woman (CEC ‘Saint-Esprit’ from AMP-MCV).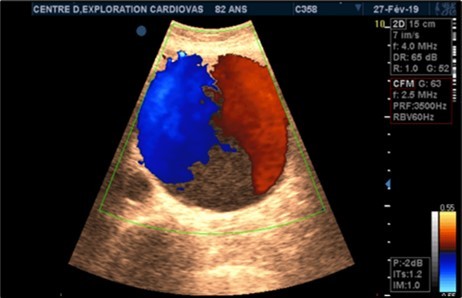
Arterial Doppler Ultrasound Data: Aneurysm Extensions
The aneurysms had extensions to other arteries in 21 patients (65.63%) distributed as follows: superior mesenteric artery (11.76%) (Figure 3) and (Figure 4), celiac trunk (14.23%), iliac arteries (57.14%) femoral arteries (19.05%). (Figure 5) and (Figure 6)
Figure 5.Subrenal AAA of 5.38 cm partially thrombosed in half in an 80-year-old subject (CHU-Kara).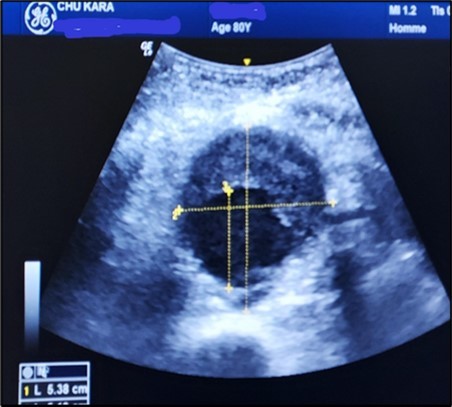
Figure 6.AAA extension with extension to the common iliac artery in a 61-year-old patient (CHU-Kara)
Patient Outcome
Of the 32 patients surveyed, 26 patients (81.25%) died, and 3 patients were lost to follow-up. Only one patient was able to undergo surgery in Europe. Death occurred by sudden death at home according to the parents in 19 patients (73.08%); in the other 7 patients (26.92%), death occurred as a result of ischemic complications downstream of the aneurysm.
Discussion
Sociodemographic Data
We selected 32 cases (4.39%) of AAA in patients at very high cardiovascular risk and especially who had downstream ischemic complications. There may be a recruitment bias compared with screening in an asymptomatic population. This bias may explain our high mean age of 73.50 9.07 years compared to that reported by Papa Diengi (64years) in 2015 in Senegal 11. Whether it is this study in Togo, or that of Papa Diengi in Senegal, or that of Kunishige in Japan male predominance has been reported 11, 12. It can be noted that male sex is a risk factor for AAA.
Circumstances of Discovery
Usually, AAAs are discovered incidentally, during a paraclinical examination such as abdominal ultrasound, arterial Doppler ultrasound, abdominal CT scan or abdominal MRI 8. But AAAs in our study had been diagnosed for arterial Doppler explorations of MI ischemia in 78.75% of cases. This proves that systematic screening for AAA in elderly subjects is almost absent in our countries. We had 2 cases of suspected AAA in front of a flailing abdominal mass. Hence the need to properly examine patients with risk factors for AAA. We did not have a case of rupture as a circumstance of discovery since it was a study of Doppler ultrasound series of diagnosis and not a resuscitation unit.
Location of AAAs
In 84.4% of patients, the AAA was subrenal. These results corroborate those of Papa Diengi in Senegal who found 63.5% of AAA under the kidney 13 and those of Baleato who reported 90% of cases of AAA under the kidney in France 14. Apparently, AAAs are preferentially located in the subrenal area. This could be due to the physiological and anatomical loss of elastic elements of the aorta as one moves away from the heart.
AH was associated with the suprarenal location with a p=0.01 and dyslipidemia with the subrenal location with a p=0.04. Baleato et al had made the same findings 14. There is evidence that hypertensive patients tend to have suprarenal AAAs. This could be due to the higher pressure on segment 4 of the aorta in hypertensive patients compared to segment 5.
Aneurysm Shape
AAAs were fusiform in 23 (71.9%) of our patients. Saccular AAAs were present in nine patients (28.1%). Beredji had reported 72.5% of cases of fusiform aneurysms and 27.5% of saccular aneurysms 15. Fusiform AAAs appear to be more frequent in the literature. The association between hypertension and fusiform AAA was significant in this study with a p= 0.01, suggesting that hypertensive patients have a tendency to present a saccular aneurysm.
Aneurysm Size
Patients with AAAs larger than 5 cm accounted for 78%, including 34.38% with sizes larger than 7 cm. There was no significant association between age and AAA size. Gender was not associated with AAA size; however, there was a significant association between diabetes and aneurysms larger than 5 cm in our series. Diabetic patients are at high cardiovascular risk and have significant arterial remodeling in the intima; therefore, more extensive and accelerated embrittlement of the aortic intimal walls.
Intra-Aneurysmal and Downstream Thrombotic Complications
Intraaneurysmal thrombotic complications were present in 75% of cases in this study. Kunishige reported 70% thrombotic complications in Japan 12.
Arterial occlusion downstream of the aneurysm was found in 73.33% of cases in this study. Faries reported 80% of occlusions downstream of AAAs in the USA 16. All these thrombo-embolic complications show the serious nature of an AAA independently of the risk of rupture.
Patient Outcome
The mortality rate in our study was 81.25%. Basnyat in Wales 10, Hoffman in the United Kingdom 17, reported respectively a hospital mortality rate of 50% and 50 to 70%. This excess mortality in our study is multifactorial: 3/4 of our patients already had ischemia downstream of the AAA, which is life threatening in the short and medium term, independently of the local complications of the AAA; the average size of the AAA was greater than 5 cm in more than 78% of cases, with an increased risk of rupture; the absence of a technical platform to operate on these AAAs, despite the presence of vascular surgeons. Indeed, the risk of rupture increases exponentially with the size 18 of the AAA. AAAs should be operated on when they reach 5 cm in size or increase in size by more than 0.5 cm per 6 months 19.
Conclusion
Abdominal aortic aneurysms are serious and fatal pathologies. They are often asymptomatic, poorly detected and poorly documented, even in populations at risk in Africa. In this study, the circumstances of discovery of AAAs are dominated by downstream complications such as ischemia at the gangrene stage most often. The clinical picture is confused with stage III and IV PIA. These patients could be qualified as incidental findings in our context. Because the dreaded complication of AAA remains rupture with sudden death. This is why early detection is necessary, especially in populations at risk.
Aorto-iliac Doppler ultrasonography is a very important, inexpensive, non-invasive, and reproducible tool for the positive and lesion diagnosis of abdominal aortic aneurysms with a sensitivity and specificity close to 100% in well-trained operators. It is also the tool of recommendation for early detection in order to institute early management or monitoring to avoid sudden rupture of very poor prognosis even in developed countries.
The site of AAA predominate in the subrenal aorta. Aneurysms are fusiform, thrombosed and present extensions to other arteries.
The prevention of aortic aneurysm in patients at risk requires a good control of cardiovascular risk factors and a systematic screening from 60 years old as well as the systematic realization of autopsies. Other studies could focus on mass screening for AAA throughout the Togolese territory in at-risk populations.
References
- 1.Vautrin E, Thony F, Chavanon O, H, PJ-P Baguet.2012).Aortic dissection spread to the renal arteries: Interest of the renal volumetry after angioplasty. Annales de Cardiologie et d’Angéiologie 61:-203–208.
- 2.Fabre O, Guesnier L, Renaut C, Gautier L, Geronimi H et al. (2005) Prise en charge actuelle des dissections aortiques de type A. Traitement chirurgical et traitement des syndromes de malperfusion. , Annales de Cardiologie et d’Angéiologie 54, 332-338.
- 3.Hoffmann B, Um P, Bessman E S, Ding R, Kelen G D et al. (2009) Routine screening for asymptomatic abdominal aortic aneurysm in high-risk patients is not recommended in emergency departments that are frequently crowded. , ACAD EMERG MED 16(11), 1242-1250.
- 4.Costantino T G, Bruno E C, Handly N, Dean A J. (2005) Accuracy of emergency medicine ultrasound in the evaluation of abdominal aortic aneurysm. , J Emerg Med.; 29, 455-460.
- 5.H L Codjo, M Savi De Tove K, Djochou M J C, Dohou S H M, D M Houenassi. (2015) Evaluation échographique du diamètre de l’aorte abdominale dans une population a risqué d’anévrysme en milieu hospitalier à Parakou Benin. Cardiologie Tropicale. 3(30), 87.
- 6.Kieffer E. (1990) Les anévrysmes de l’aorte sous-rénale. in Collection « Actualités de Chirurgie vasculaire ». Editions AERCV , Paris 7(102), 119.
- 7.Kuhn M, Bonnin R L, Davey M J, Rowland J L, Langlois S L. (2000) Emergency department ultrasound scanning for abdominal aortic aneurysm: accessible, accurate, and advantageous. , Ann Emerg Med 36, 23-219.
- 8.Hoffman M, Avellone J C, Plecha F R, Rhodes R S, Donovan D L et al. (2002) Operation for ruptured abdominal aortic aneurysms: a community-wide experience. , Surgyre;(91): 4, 602.
- 9.Newitt M P. (1989) Prognosis of abdominal aortic aneurysms. A population based study. New England. , J. Med 5(321), 1014.
- 10.Santé Haute Autorité de. (2012) Recommandation en santé publique. Dépistage et prévention des anévrismes del’aorte abdominale.https://www.has-sante.fr/jcms/c_1358519/fr/pertinence-de-la-mise-en-place-d-un-programme-de-depistage-des-anevrismes-de-l-aorte-abdominale-en-france.
- 11.P A Dieng, M S Diop, P S Ba, Diatta S, Gaye M et al. (2015) Anévrismes de l’aorte abdominale sous-rénale rompus: aspects chirurgicaux à Dakar: à propos de 6 observations. , Pan African Medical Journal 11(21), 241-57.
- 12.KunishigeH IshibashiY, Kawasaki M. (2013) Risk factors affecting survival after surgical repair of ruptured abdominal aortic aneurysm. Ann Vasc Dis. 6(3) :. 631-636.
- 13.A D Papa, Diop N, Diarra O, A G Ciss, Ndiaye A et al. (2007) Chirurgie des anévrysmes de l’aorte abdominale sous rénale à dakar. , Ann. Afr. Chir. Thor. Cardiovasc 2(1), 53-58.
- 14.Baleato S, Bierry G, Garcia-Figueiras R. (2008) Anévrismes artériels ; revue des différents territoires. Journées françaises de radiologie. , Paris.: 24-28.
- 15.Beregi J P, Prat A, Gaxotte V, Delomez M, McFadden E P. (2000) Endovascular treatment for dissection of the descending aorta. , Lancet 5(356), 482-3.
- 16.Faries P L, Brener B J, Connelly T L, Katzen B T, Briggs V L et al. (2002) A multicenter experience with the Talent endovascular graft for the treatment of abdominal aortic aneurysms. , J Vasc Surg 35(6), 1123-8.
- 17.Kieffer E. (1990) Les anévrysmes de l’aorte sous-rénale. in Collection « Actualités de Chirurgie vasculaire ». Editions AERCV, Paris . 7(102) : 119.
