Maternal Behavior Affects Child’s Attachment-Related Cortisol Stress Response
Abstract
Background
Mothers with a history of childhood maltreatment (CM) are likely to transmit their own experiences to the next generation. This is highly influenced by the quality of maternal behavior that enables to buffer infant’s hypothalamic-pituitary-adrenal (HPA) axis response to stress. From a transgenerational perspective the research question is, if infant’s cortisol stress response is influenced by maternal CM experiences or rather by the behavioral pathways during the first year of life.
Methods
53 mother-child-dyads were measured at 12 months of infant’s age in a laboratory visit assessing the maternal quality of interactive behavior using the Atypical Maternal Behavior Instrument for Assessment and Classification (AMBIANCE) measured during the strange situation procedure (SSP). Maltreatment experiences were assessed using the childhood trauma questionnaire (CTQ). Salivary cortisol of mother and infant were collected prior to and directly, 15 and 30 minutes after the SSP.
Results
Infants of disruptive mothers showed a significant increase in cortisol (F (3; 147) = 2.897, p= 0.048) after the SSP compared to infants of mothers with sensitive caregiving. Maternal CM did not influence the infant’s cortisol stress response due to the SSP. However maternal cortisol response was altered by trend due to CM (F (1.392; 71.008) = 3.157, p= 0.066).
Conclusions
Our data indicate that infant’s cortisol stress reactivity is influenced by the quality of maternal behavior and not by the transgenerational transmission of maternal CM experiences per se. These findings implicate that helping mothers to improve their caregiving behavior may help to improve stress-reactivity of their infant.
Author Contributions
Academic Editor: Baoman Li, China Medical University, China.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2019 Franziska Köhler-Dauner, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
Childhood maltreatment (CM) is considered as a significant risk factor for detrimental development, in particular when it occurs early in life 1, 2. Individuals with experiences of CM are at higher risk for long-term serious health consequences on 1 psychological level (e.g. elevated risk for psychiatric disorders 3, increased sensitivity for everyday stressors 4 and perception of stress 4 and 2 physical level (e.g. more medical diagnoses 5 and lower expectancy of life 6. CM has been shown to be transmitted to the next generation via behavioral (e.g., quality of parenting) 7 and presumably via biological pathways 8, 9, 10.
Dysregulation of the hypothalamic-pituitary-adrenal (HPA) axis has been widely shown in the context of CM. The HPA axis, considered as the body’s major glandotrop stress system, is associated with increased vulnerability to psychological and physical diseases 11. Stress is regulated via the HPA axis, which exerts an adaptive stress response of the organism. The stress response initiates a biochemical cascade where corticotropin-releasing hormone (CRH) is delivered from the hypothalamus. The delivery of CRH triggers the flow of the adrenocorticotropin-releasing hormone (ACTH) produced by the anterior pituitary, which, on the other hand, activates the release of glucocorticoids from the adrenal cortex 1, 12. During upcoming stress, the HPA axis increases the production of cortisol to offer the capacity to mobilize energy or priming the body for similar stressors in the future 13. Cortisol increase reaches a peak between 20 to 40 minutes after a stressor and falls back to the baseline when the stressful situation is resolved 14, with peak levels of infant´s cortisol within 20 to 40 minutes after a stressor 15. Functional stability of the HPA axis is not definitively developed at birth but infant´s cortisol baseline secretion and peak percentage increases week by week until the age of 1 year. In 1 year old infant, the HPA axis has become socially regulated which protect infant´s development from detrimental effects of excessive exposure to glucocorticoids 16, 17.
CM and HPA Reactivity
Specifically, the stress of experiencing CM has been linked to HPA dysregulation during the period of maltreatment as well as until adulthood 17. Ongoing stress and stress of which the individual is not able to effectively cope with may change the sensitivity of the glucocorticoid receptors, resulting in alterations in cortisol delivery and affects other brain structures such as the hippocampus 18. Several studies have examined the effect of CM showing that individuals with CM experiences demonstrate dysregulated cortisol levels measured in urine, blood and saliva 19, 20. Morris and colleagues (2012) showed that adults, who were maltreated as children, tend to exhibit lower levels of cortisol in the evening 21 and blunted responsiveness to psychosocial challenges 22. In contrast, Heim et al. identified increased cortisol responsiveness to challenge in context of CM 23. Especially in maltreated children, the research findings are less clear 24 revealing increased, decreased or similar patterns of cortisol reactivity compared to a non-maltreated control group 17. In detail, Cicchetti and Rogosch (2001) and Bruce and colleagues (2009) detected that physically abused children showed lower salivary cortisol levels and flattened diurnal slope 25, 26 indicating a down-regulation of the HPA axis in response to initial cortisol increasing the context of CM 18. Both, boys and girls with CM experiences showed attenuated cortisol stress responses in stressful situations 24, 27, 28. In addition, studies of toddlers living in Russia and Romania showed decreased morning cortisol levels and no diurnal salivary cortisol decrease throughout the day 29. This has also been shown to be apparent among preschoolers in foster care, of which 35% showed blunted cortisol diurnal rhythm 30. In contrast to these findings, De Bellis et al. (1999) showed that children with experiences of CM and a consecutive posttraumatic stress disorder had increased cortisol levels 19. Furthermore, it has been shown that cortisol levels increased with the severity and duration of CM 12. Longitudinal studies of HPA axis regulation in maltreated individuals are rare 24. Nevertheless, one study realized a longitudinal HPA axis follow-up from childhood over adolescence to adulthood in maltreated girls. They reported high cortisol levels in childhood, attenuated cortisol in adolescence and exhibited low cortisol levels in adulthood 31, which suggest a shift from hypocortisolism in childhood to receptor down-regulation in adulthood in response to chronic stress 28.
Recently, a growing research interest has been focused on the biological correlates of trans-generational transmission of CM showing that maternal child abuse results in disturbances in the HPA axis in their offspring. Brand and colleagues (2010) detected that children of mothers with CM showed lower baseline cortisol compared to children of mothers without CM. Additionally, children of mothers of a combination of both, a history of early maternal abuse and comorbid post-traumatic stress disorder, were associated with greater increases in child’s cortisol levels 32. Fisher et al. (2007) observed in a 25 - year longitudinal study lower morning cortisol and modified cortisol release during the day in children of mothers´ with experiences of CM compared to children without CM experiences of their mothers 33.
The transmission of HPA axis dysregulation due to CM into the next generation was confirmed by Yehuda and colleagues (2005) who reported, that offspring of mothers with traumatic experiences in adulthood and PTSD showed significantly lower cortisol levels in saliva and blood compared to a control group 34.
Quality of Caregiving and HPA Axis Reactivity
In childhood sensitive, warm and responsive caregiving plays an equivalent role in successful child’s behavioral and physiological regulation outcomes 35. High quality of caregiving is critical in reducing young infants´ distress in situations of emotional arousal and may buffer infant´s HPA axis response during stress 18. Extensive evidence of research findings indicates that excessive or prolonged activation of stress response in the first years of infants' life predicts later psychophysiological healing. It is particularly important to differ between types of stress: positive stress, tolerable stress and toxic stress. Infants could cope with both types of stress like positive stress as well as tolerable stress with moderate, short-lived stress responses respectively stress with potential to negatively affect but over limited time periods. Especially toxic stress in childhood revealed correlations with emotional behavior, health across lifespan and promotes damaging effects on learning 36, 37. Recently, physiological parameters have been used to confirm behavioral observations and to enable a more basal understanding of the relation between behavioral and physiological systems. The HPA axis stress response has been used to study physiological stress responsiveness to arousing or stressful situations in humans and animals 38, 39. The adrenocortical system seems to be particularly responsive to aversive or stressful situations that convey novelty or uncertainty accompanied by negative emotions 48. The earliest years in infants´ life are well known to be a critical window for disturbances in stress regulation processes. Both, sensitive and intrusive maternal interacting behaviors in free play and stressful situations have been associated with alterations of infants´ stress regulation outcomes 39. Especially the daily quality of caregiving may affect the development of infant’s stress regulatory capacities 17, 40, 53. Gunnar et al. (1992) found associations between the quality of caregiving and the adrenocortical response of the infant during a paradigm of separation of mother and infant. Their findings suggested that caregiving might buffer the adrenocortical response of the infant in stressful situations 41. These findings were replicated by Enlow et al. (2014), who found that higher maternal insensitivity was associated with higher cortisol release during a stressful laboratory paradigm 42. This was confirmed by Haley et al. 2003 who reported that infants of more responsive parents showed greater regulation like lower peaks of cortisol in stress situations in their HPA stress response than infants of less responsive parents 43. In contrast Nachmias et al. (1996) showed no cortisol increase during an experimental setting of live clown in children of more sensitive parents compared to lower sensitive caregiving 44. On the other hand a high quality of caregiving is supposed enhance secure attachment between child and caregiver 55, and children who are securely attached showed no anomalous elevation of cortisol reaction when the attachment figure was present 46, 47, 48. Cicchetti et al. suggested sensitive caregiving to play a major role on infants´ HPA axis regulation 49. However, there are only few comparative studies focusing on both, maternal behavior and maltreatment and their effects on infants´ HPA axis reactivity during stressful situations 50. For example Martinez-Torteya (2014) evaluated the effect of maternal parenting on infant’s biobehavioral regulation among maternal CM 51. They found no direct influence of infants´ physiological or behavioral responses but positive associations between maternal interaction quality and infant´s cortisol reactivity to stressors 51. Our investigations may shed light on the influence of maternal CM and the quality of caregiving on infants´ stress response one year after birth. Especially, the question of a transgenerational transmission of CM on behavior level should be clarified. Therefore, we aimed to differentiate maternal CM and caregiving on child’s cortisol stress reactivity and measured salivary cortisol in the mothers-child-dyad during the SSP. We hypothesized 1. that maternal cortisol response during SSP differs between mothers´ with and without CM and 2. that the cortisol response of the child will be transmitted by interacting behavior of the mothers´ in the first year of life and not by the history of maternal CM.
Material and Methods
Participants and Study Design
TRANS-GEN is an interdisciplinary study consortium investigating in a prospective approach the pathways leading to resilience or vulnerability in the transgenerational transmission of childhood maltreatment (CM) by focusing psychological, biological and social factors.
The study was funded by the Federal Ministry of Education and Research and was approved by the Ethics Committee of Ulm University. After recruiting in the maternity unit of the Ulm University Hospital all mother-infant dyads were followed up twice: 3 months (t1) and 12 months (t2) after birth.
Participants
Since October 2013, 533 mother-infant-dyads were being recruited in the women’s hospital of the University Hospital of Ulm 1-6 days after parturition and were willing to complete the screening interview (t0). Inclusion criteria were age >18, over 37 weeks of pregnancy, sufficient knowledge of the German language, no complications during parturition or health problems of mother and/or infant as well as no current drug consumption or a history of severe psychiatric disorders or current infections. 240 mother-infant-dyads could be invited for a follow-up 3 months (t1) after birth in both laboratory as well as in home visit and 158 mother-infant-dyads participated in a further laboratory and home visit around 12 months of infant’s age (t2).
All mother-child-dyads were asked to collect salvia of 4 measurement points. The sample for the following analyses included 53 mother-infant-dyads. Missing data sets were due to the lack of agreement for collecting biological samples of 1 cases of mothers-child-dyads. In addition, 104 of mothers´ and 106 of infant´s collected salvia had no or insufficient amount of saliva for each data point, before (baseline before SSP), directly after (+1 min.) 15 minutes (+15 min.) and 30 minutes (+30 min.) the SSP. For final analysis, we included only mothers and their infants with a complete data set of 4 measurements. Cortisol levels of two infants could not be analyzed due to low amount of saliva. Therefore, we considered complete data sets of 53 mothers and 51 infants for final analyses (complete data sets means no missing of any of each 4 measurement points).
Mothers’ age at time of measurement (t2) was between 21 and 43 years (mean 33.72 years (SD 4.47 years)). Maternal body mass index (BMI) was between 17.26 and 31.83 (mean 24.35 (SD 3.82). 79.2% of the mothers reported to be married or living in a partnership and 88.7% of all mothers had German citizenship. Mother’s level of education at t2 in comparison to the educational background of the German population showed, that 56.6% had a grammar school degree, 11.3% a secondary school degree, 22.6% a basic secondary school degree and 7.5 % no school diploma. Furthermore, 20.8% of the mothers had medical risk factors e.g. chronic disease, high blood pressure or allergies. 30 male and 21 female infants were investigated in laboratory visit. All mother-infant-dyads were invited around 12 months of infant´s age (12.0 ± 0.1 months) (mean 12.43 years (SD 0.80 years)) (Table 1).
Procedure
Mothers were screened for CM using the CTQ 52. Experiences of emotional abuse, physical abuse, emotional neglect, physical neglect and sexual abuse are assessed with five items each rated on a five-point Likert scale. The CTQ subscale scores range from 5 to 25 and the total scores from 25 to 125. The sum score over all 25 items was calculated as a cumulative measure from “none” maltreatment experiences (25 points) up to “minimal” till “extreme” maltreatment load 53. All ratings were summed up to a sum score. Mothers´ with a higher sum score than 25 were classified as CM+.
All mother infant-dyads were invited for a laboratory visit at 12 months of infant’s age (t2). Due to circadian rhythm of cortisol levels, all mother-child-dyads were assessed from 10.00 am to 1.00 pm. After a short introduction explaining the procedure of the investigation, mothers and infants were asked to have a resting phase of 15-20 minutes before sampling of the first saliva probes (baseline before SSP) prior to the beginning of the SSP. Further saliva samples were collected directly (+1 min. after SSP), 15 minutes (+15 min. after SSP) and 30 minutes (+30 min. after SSP) after the SSP while mothers were asked to play with their infant and relax by using toys. The implementation of the SSP was based on the standardized SSP protocol including 7: (e1) mother and infant were alone in the room with the infant exploring the room and the mother sitting on a chair, (e2) first encounter and interaction with the stranger, (e3) mother went out of the room (first separation), (e4) mother came back after a time period ranging from 30 seconds and 3 minutes dependent on child´s irritation and reaction of being separated from the mother (stranger left the room while reunion), 5 mother left the room for the second time while infant is alone in the room (second separation), (e6) the stranger came back instead of the mother, (e7) the mother came into the room (second reunion) while the stranger went out of the room 54.
Ambiance Measure
For measuring the quality of maternal interactive behavior we videotaped mother-infant interactions during the SSP analyzed by the “Atypical Maternal Behavior Instrument for Assessment and Classification (AMBIANCE)” (AMBIANCE;55). The AMBIANCE is based on the theory of Main and Hesse (1990) and has been developed by Lyons-Ruth and colleagues and assesses anomalous parental behavior of mothers´ during interactions with their infant 55, 56, 57.
Additionally to the concepts of frightened and frightening behaviors and dissociated parental states described by Main and Hesse 56 AMBIANCE considers profound disruptions in mother – infant interaction as well as behaviors that are physically or emotionally withdrawn 58. To assess the quality of interacting behavior AMBIANCE codes disrupted maternal behaviors on five dimensions: affective communication errors, role/boundary confusion, disorganized/ disoriented behaviors, negative/intrusive behavior, and withdrawal. Each dimension is coded on a 7-point scale as well as an overall score of the level of disruption. The frequency and intensity of all disrupted behaviors mothers´ displayed in the course of the interaction with their infant resulted in the level of maternal disrupted communication. Maternal disrupted communication coded up to 4 has been considered “not-disrupted” whereas a level from 5 to 7 is considered “disrupted”. Therefore, mothers with maternal communication below 5 were coded as “not-disrupted” whereas mothers coded from 5 till 7 were coded as “disrupted” mothers. All videotaped play sessions were scored by a single coder, who was blind to all other data of the mother-infant-dyads. This coder was trained by and reliable with the original developers of the AMBIANCE 55.
Saliva Sampling
For saliva sampling, SalivaBio’s Children’s Swabs (SCS) (Salimetrics, State College, USA) were used by following the standard procedures. Therefore, SCS were placed into the infant’s and mothers´ mouths for about 30 seconds. Thereafter, swabs were stored in storage tubes on ice at -20°C. Upon completion of t4, tubes were centrifugalized at 4°C/ rpm and aliquots were stored at -80° C. Cortisol levels (µg/dL) were analyzed by C. Kirschbaum (Technical University of Dresden, Germany). The salivary concentrations were measured using chemiluminescence immunoassay with high sensitivity (IBL International, Hamburg, Germany). The intra and interassay coefficients for cortisol were below 8%.
Statistical Analyses
We conducted statistical analyses using Statistical Package for the Social Sciences version 23.0 (SSP Inc., Chicago, IL). Statistically significant was set at p<.05. Normal distribution of data was tested by non-parametric Kolmogorov-Smirnov test. Since data were normally distributed, data were analyzed as follows: ANCOVA for repeated measures was calculated for each of the cortisol values between subject (group: “CM-” / “CM+”; “not-disrupted” / “disrupted” maternal behavior; mother, infant) and within subjects (for episode “baseline before SSP” to “+30 min.”). Greenhouse-Geisser as well as Sphericity assumed correction for repeated measures were applied. To test differences between the groups within each measurement we used unpaired t-tests. Infant sex, age of the mother at birth as well as the body mass index of the mother were controlled as covariates (Table 1).
Results
Descriptive Analyses
Descriptive statistics are shown in Table 1. Twenty-eight of 53 mothers were grouped as CM+ and 21 showed “disrupted” maternal behavior (Table 2 and Table 3). No significant differences were detected between the CM- and CM+ mothers as well as “non-disrupted” and the “disrupted” maternal behavior group concerning infant’s sex, mother’s age at birth and body mass indexes and thus were not considered for further analyses. There were also no significant differences for maternal interacting behavior focusing maternal education as well as their current marriage status.
Table 1. Descriptive analyses of covariates| N | Min | Max | Mean | SD | |
| Mother’s age at birth | 53 | 21 | 43 | 33.72 | 4.47 |
| Body mass indexes | 53 | 17.26 | 31.83 | 24.35 | 3.82 |
| Infants´ age | 53 | 11 | 14 | 12.43 | 0.80 |
| N | Range | Min. | Max. | Mean | Std. Deviation | Variance | ||
| Statistic | Statistic | Statistic | Statistic | Statistic | Std. Error | Statistic | Statistic | |
| AMBIANCE overall score | 53 | 5 | 1 | 6 | 3,96 | ,166 | 1,208 | 1,460 |
| Valid N (listwise) | 53 | |||||||
| Frequency | Percent | Valid Percent | Cumulative Percent | |||||
| AMBIANCE overall score | ´non-disruptive´ | 32 | 60,4 | 60,4 | 60,4 | |||
| ´disruptive´ | 31 | 39,6 | 39,6 | 100,0 | ||||
| total | 53 | 100,0 | 100,0 | |||||
| N | Range | Min. | Mean | Std. Deviation | ||
| Statistic | Statistic | Statistic | Statistic | Std. Error | Statistic | |
| Maternal CM | 53 | 70 | 1 | 36,53 | 2,043 | 14,871 |
| Valid N (listwise) | 53 | |||||
| Frequency | Valid Percent | |||||
| Maternal CM | CM- | 25 | 47,2 | |||
| CM+ | 28 | 52,8 | ||||
| total | 53 | 100,0 | ||||
Maternal Experiences of CM and Cortisol Reactivity in Mothers and their Infant
Focusing maternal HPA axis reactivity during SSP by using ANCOVA for repeated measures, neither main time (F (1.392; 71.008) = 0.579, p= 0.503), nor group effects (F (1; 207.539) = 1.052, p= 0.310) were detectable for CM+ compared to CM-. The values showed marginal, but no significant differences for group-by-time effects (F (1.392; 71.008) = 3.157, p= 0.066) (Figure 1a).
Figure 1a.ANCOVA for repeated measures for CM and maternal cortisol reactivity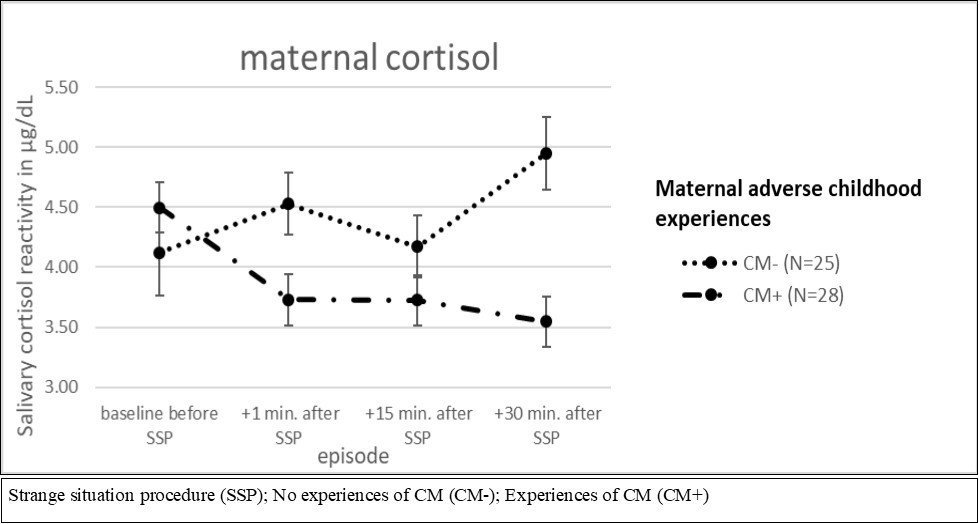
For infant´s HPA axis time effects (F (2.226; 109.096) = 14.277, p= 0.000) could be shown. For infant’s main group-by-time (F (2.226; 109.096) = 0.595, p= 0.571) or group effects (F (1; 49) = 0.740, p= 0.394), no significant results were detectable (Figure 1b).
Figure 1b.ANCOVA for repeated measures for CM and infants´ cortisol reactivity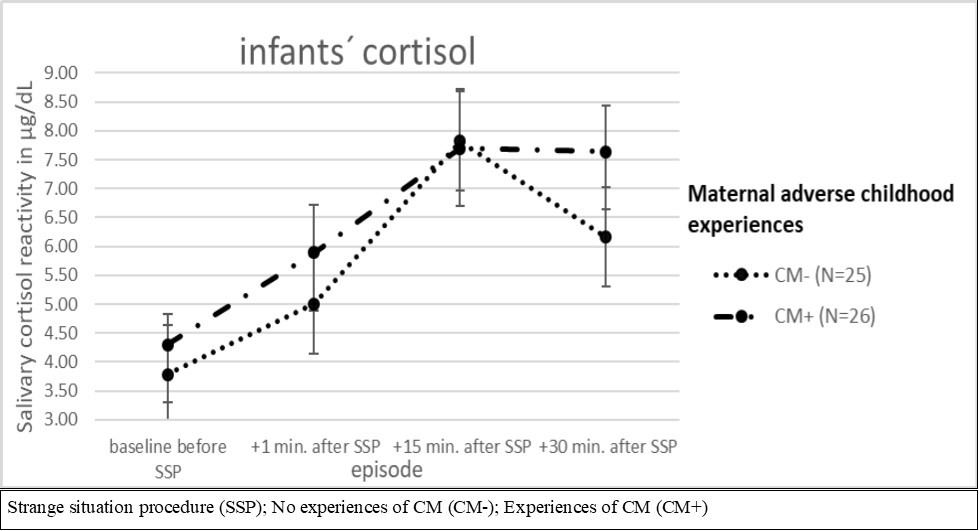
T-test analyses comparing two independent samples revealed no differences focusing maternal and infant´s cortisol values and CM for each of the measurements (baseline before SSP - +30 min.).
Maternal interacting quality and cortisol reactivity in mothers and their infant
Using ANCOVA for repeated measures the saliva cortisol in mothers relative to maternal interacting behavior showed neither main time (F (1.398; 71.305) = 0.663, p= 0.466), group-by-time (F (1.398; 71.305) = 1.097, p= 0.320) nor group effects (F (1; 51) = 0.053, p= 0.819) (Figure 2a).
Figure 2a.ANCOVA for repeated measures for AMBIANCE overall score of “non-disruptive´ vs. ´disruptive´ behavior and maternal cortisol reactivity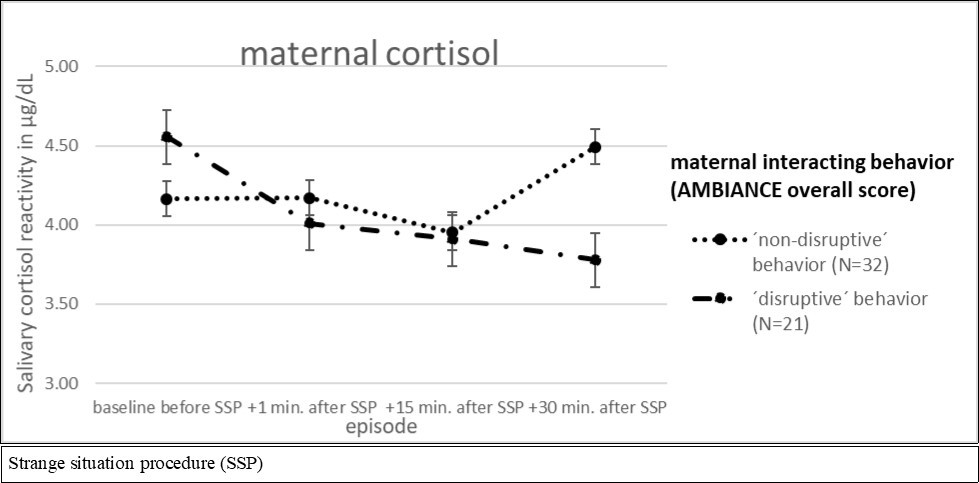
Saliva cortisol in their infants, however, showed significant time (F (3; 147) = 16.472, p= 0.000) as well as significant main group-by-time effects (F (3; 147) = 2.897, p= 0.048). For main group effects no significant differences could be shown (F (1; 49) = 1.664, p= 0.203) (Figure 2b).
Figure 2b.ANCOVA for repeated measures for AMBIANCE overall score of “non-disruptive´ vs. ´disruptive´ behavior and infants´ cortisol reactivity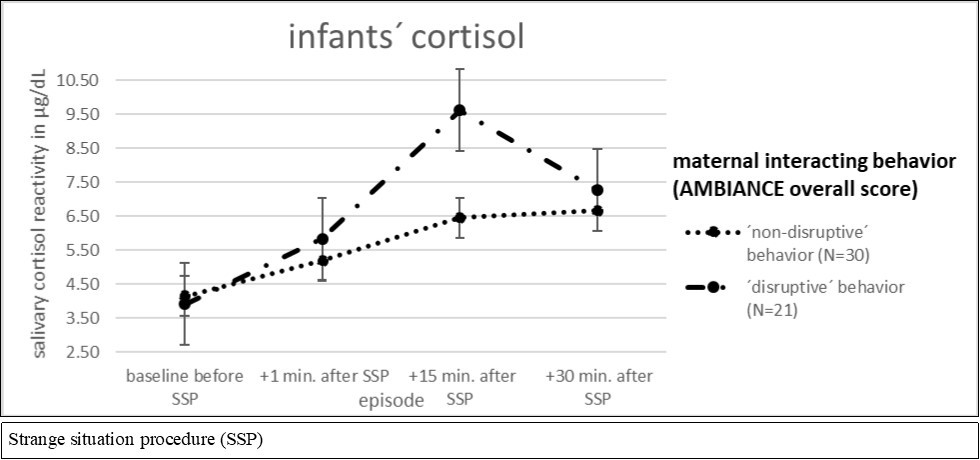
T-test analyses comparing two independent samples showed no differences for maternal cortisol levels focusing maternal interacting behavior. For infants cortisol values differences could be shown for the measurement 15 minutes after SSP (+15 min.) (t(49) = -2.108, p = 0.040). Infants of mothers with lower sensitive caregiving showed significant higher cortisol values (M = 6.46, SD = 3.525)than infants of mothers with a higher sensitive interacting behavior (M = 9.613, SD = 7.051).
Behavior and Infants´ Cortisol Reactivity
Correlation Between CM and the Quality of Maternal Caregiving
Pearson correlation analyses between the CTQ sum score of maternal maltreatment and interacting behavior measured by the AMBIANCE global score revealed no significant associations (r(53)=.080, p=0.567) (Table 4).
Table 4. Pearson Correlations between AMBIANCE overall score and CTQ overall score| AMBIANCE overall score | CTQ overall score | ||
| AMBIANCE overall score | Pearson Correlation | 1 | ,131 |
| Sig. (2-tailed) | ,348 | ||
| N | 53 | 53 | |
| CTQ overall score | Pearson Correlation | ,131 | 1 |
| Sig. (2-tailed) | ,348 | ||
| N | 53 | 53 |
Discussion
Our findings reveal differences in the cortisol stress responses of mothers and infants in relation to maternal CM and the quality of interacting behavior. Mothers with a history of CM showed no significant, but by trend differences in their cortisol stress response compared to mothers without CM. Transgenerational effects on cortisol stress reactivity in the child were only present when mothers showed disruptive maternal behavior towards the child, but not when mothers have reported maltreatment in their history
Infants Cortisol Reactivity Depending on Maternal CM and Interacting Quality
Focusing the infant’s cortisol response in association to maternal CM we could not find any differences between both groups of infants with and without maternal CM. This is in line with Martinez-Torteya et al. (2014) who found no direct association between maternal CM and infant’s cortisol reactivity after stressors 51. However, two longitudinal studies of Fisher et al. (2007) and Brand et al. (2010) described differences in baseline cortisol levels as well as modified cortisol release during the day in infants of mothers´ with experiences of CM compared to infants without maternal CM 32, 33. The divergence between our results and that of Fisher et al. (2007) and Brand et al. (2010) may be explained by the low severity of maternal CM load in our sample. Fisher et al. (2007) and Brand et al. (2010) measured a sample with a range from low to high maltreatment. In contrast to that, our sample showed only low to moderate maltreatment load. We might consider that severity of maternal CM load affects the transmission of own experiences of CM into the next generation. This is in accordance with Alink et al. (2012) who showed that the risk of dysregulation in cortisol levels of mothers with CM increased in dependence to the severity and the duration of CM 12. Therefore, we conclude that lower CM experiences in our sample may lead to a lower risk for transgenerational transmission of HPA axis dysregulations.
Interestingly, our data showed that maternal interacting quality seems to be a main factor that influence the infant´s cortisol stress regulation. Infants of mothers´ with disruptive interacting behavior showed a significant peak of cortisol level after 15 minutes compared to infants of mothers with sensitive interacting behavior. This indicates that enhanced sensitive caregiving resulted in lower infant’s cortisol stress response. The results are in line with previous studies showing that a higher quality of caregiving resulted in a reduced infant’s cortisol stress response 17, 46, 51. Especially during the first years of life, infants need to manage the challenging transition from external regulation of affect and internal arousal to rising levels of psychobiological regulation 59. In attachment as well as developmental theory it is suggested that the quality of parental interacting behavior as well as the relationship between infants and parents are an important factor for the development of infant’s emotional and physiological regulatory strategies 47, 59.
The SSP is conceived as a stressful procedure in which the infant is stressed by two short separations from the caregiver. Several studies confirmed that the quality of maternal interacting behavior may buffer infants´ distress in situation of emotional arousal 60. A stable and reliable relationship between mother and infant based on maternal sensitive interacting behavior might reduce infants´ distress due to the SSP and stabilize the infant at the time of the reunion with the caregiver. Lower sensitive and supportive parenting (e.g. like frightening and anxious interaction behavior) might constrain or reduce the ability of physiological regulation, reflected by a dysregulation of infants’ cortisol stress response 40, 61.
Maternal Cortisol Reactivity in Relation to CM and Their Interacting Quality
Mothers´ with a history of CM showed no significant but marginal differences in course of their cortisol levels compared to mothers without CM. However mothers with a history of CM started with a lower but not significant differences in cortisol baseline than mothers without CM. This was also found by Trickett et al., (2010) who showed in a longitudinal design that adults maltreated in childhood showed lower cortisol baselines than adults without CM 31. Our findings show that the severity of experiencing CM may affect the regulation of the HPA axis until adulthood which is confirmed by data of Tarullo and Gunnar 17. One potential reason might be the moderate to low severity of CM load in our sample. Looking at maternal interacting quality there were no significant differences in the cortisol responses between disruptive and non-disruptive mothers. It seems that focusing differences in maternal interacting behavior in detail may not alter the cortisol reaction in mothers 21.
In summary, we did not find any cortisol-related transmission effects of CM to the next generation, however, maternal behavior seemed to play a crucial role in this context. This is a particularly important and encouraging finding with regard to the cycle of maltreatment and the fact that many parents are concerned to transmit their own experiences to their infants. Especially for parents with CM, the former experience of maltreatment was not as crucial like maternal daily interactions and the relationship with their infant. Inadequate or anomalous maternal interacting behavior may aggravate infants´ stress regulatory strategies, which could be a risk for later stress-related mental and physical burden linked to increased stress vulnerability and impaired emotion regulation 62, 63. Our results indicate that especially the daily experienced interacting quality by their mothers is relevant for the development of infant´s HPA stress regulation in the first year of life.
Therefore the findings of our analyses indicate both, protection and risk factors. On the one hand, maternal CM experience has been proven to affect cortisol stress response in adulthood. Especially mothers with CM experiences may be particularly vulnerable for dysregulated HPA axis. On the other hand, we were able to show that CM experience did not directly burden the next generation in terms of their stress regulation capacities. However, daily interactions between mother and infant contribute to the development of infants´ resilient stress responsiveness.
Limitations
Limitations of this study were saliva acquisition problems due to mothers´ and infants´ limited amounts. This resulted in the small sample size of complete data sets. Additionally, in contrast to other studies with high maltreatment load our study showed a rather low severity of CM experiences. This has to be taken into account when comparing our results with other studies with samples showing wider range of CM experiences. Demographic characteristics of our sample can be considered as one limitation since the level of maternal education was high compared to other samples with history of CM.
Conclusion
Mothers with a history of CM are likely to transmit their own experiences to the next generation. Our data show that not CM per se but the current quality of maternal caregiving plays a crucial role for the regulation of infant’s cortisol stress reactivity. We may conclude that CM experiences are transmitted to the next generation via maternal behavior. This can be considered as an encouraging result, focusing the research findings of early life stress since improvement of maternal behavior is part of most preventions and interventions focusing on parent-infant relationships 64.
References
- 1.Gonzalez A. (2013) The impact of childhood maltreatment on biological systems: Implications for clinical interventions. Doi: 10.1093/pch/18.8.415. Paediatrics & Child Health.18(8),415-418 .
- 2.Jaffee S R, Maikovich-Fong A K. (2011) Effects of chronic maltreatment and maltreatment timing on children's behavior and cognitive abilities. , Journal of Child Psychology and Psychiatry and Allied Disciplines
- 3.Afifi T O, Macmillan H L.Resilience following child maltreatment: A review of protective factors. , Canadian Journal of Psychiatry 56(5), 266-272.
- 4.Hyman S M, Paliwal P, Sinha R. (2007) Childhood maltreatment, perceived stress, and stress-related coping in recently abstinent cocaine dependent adults. Doi: 10.1037/0893-164X.21.2.233. Psychology of addictive behaviors. 21(2), 233-238.
- 5.Sachs-Ericsson N, Cromer K, Hernandez A, Kendall-Tackett K. (2009) A review of childhood abuse, health, and pain-related problems: the role of psychiatric disorders and current life stress. Doi: 10.1080/15299730802624585. , Journal of Trauma & Dissociation 10-2.
- 6.Brown D W, Anda R F, Tiemeier H, Felitti V J, Edwards V J et al. (2009) Adverse childhood experiences and the risk of premature mortality. Doi: 10.1016/j.amepre.2009.06.021. American journal of preventive medicine. 37(5), 389-396.
- 7.Köhler-Dauner F, Roder E, Krause S, Buchheim A, Gündel H. (2018) (submitted) Reduced caregiving quality measured during the strange situation procedure increases child's autonomic nervous system stress response.
- 8.Carr C P, CMS Martins, Stingel A M, Lemgruber V B, Juruena M F. (2013) The role of early life stress in adult psychiatric disorders: a systematic review according to childhood trauma subtypes. Doi: 10.1097/NMD.0000000000000049. The Journal of nervous and mental disease. 201(12), 1007-1020.
- 10.Schury K, Koenig A M, Isele D, Hulbert A L, Krause S. (2017) Alterations of hair cortisol and dehydroepiandrosterone in mother-infant-dyads with maternal childhood maltreatment. Doi: 10.1186/s12888-017-1367-2. BMC Psychiatry. 17(213)
- 11.Schury K, Kolassa I T. (2012) Biological memory of childhood maltreatment: Current knowledge and recommendations for future research. doi: 10.1111/j.1749-6632.2012.06617.x. Annals of the New York Academy of Sciences. 1262(1), 93-100.
- 12.LRA Alink, Cicchetti D, Kim J, Rogosch F A. (2012) Longitudinal associations among child maltreatment, social functioning, and cortisol regulation. Doi: 10.1037/a0024892. Development psychology 48(1), 224-236.
- 13.Sapolsky R M, Romero L M, Munck A U. (2000) How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Doi: 10.1210/er.21.1.55. Endocrine Reviews 21, 55-89.
- 14.Hunter A L, Minnis H, Wilson P. (2011) Altered stress responses in children exposed to early adversity: a systematic review of salivary cortisol studies. , Doi: 10.3109/10253890.2011.577848. Stress 14(6), 614-626.
- 15.Goldberg S, Levitan R, Leung E, Masellis M, Basile V S. (2003) Cortisol concentrations in 12- to 18-month-old infants: Stability over time, location and stressor. Doi: 10.1016/S0006-3223(03)00010-6. Biological Psychiatry. 54(7),719–726
- 16.Hostinar C E, Sullivan R M, Gunnar M R. (2014) Psychobiological mechanisms underlying the social buffering of the hypothalamic–pituitary–adrenocortical axis: A review of animal models and human studies across development. , Psychological Bulletin 140(1), 256-282.
- 17.Tarullo A R, Gunnar M R. (2006) Child maltreatment and the developing HPA axis. Doi: 10.1016/j.yhbeh.2006.06.010. Hormones and Behavior 50(4), 632-639.
- 18.Gunnar M, Vazquez D M. (2001) Low cortisol and a flattening of expected daytime rhythm: Potential indices of risk in human development. Doi: 10.1017/ S0954579401003066. Development and Psychopathology 13(3), 515-538.
- 19.De Bellis MD, Baum A S, Birmaher B, Keshavan M S, Eccard C H. (1999) Developmental traumatology, Part 1: Biological stress systems. Doi: 10.1016/S0006-3223(99)00044-X. Biological Psychiatry. 45(10),1259–1270
- 20.Gunnar M, Quevedo K. (2007) The neurobiology of stress and development. Doi: 10.1146/ annurev.psych.58.110405.085605. Annual Review of Psychology 58, 145-173.
- 21.Morris M C, Compas B E, Garber J. (2012) Relations among posttraumatic stress disorder, comorbid major depression, and HPA function: a systematic review and meta-analysis. Doi: 10.1016/j.cpr.2012.02.002. Clinical psychology review 32(4), 301-315.
- 22.Carpenter L L, Shattuck T T, Tyrka A R, Geracioti T D, Price L H. (2011) Effect of childhood physical abuse on cortisol stress response. Doi: 10.1007/s00213-010-2007-4. Psychopharmacology. 214(1),367–375
- 23.Heim C, Newport D J, Wagner D, Wilcox M M, Miller A H. (2002) The role of early adverse experience and adulthood stress in the prediction of neuroendocrine stress reactivity in women: a multiple regression analysis. Doi: 10.1002/da.10015. Depression and anxiety. 15(3), 117-125.
- 24.Doom J R, Cicchetti D, Rogosch F A. (2014) Longitudinal Patterns of Cortisol Regulation Differ in Maltreated and Nonmaltreated Children. , Journal of the American Academy of Child and Adolescent Psychiatry 53(11), 1206-1215.
- 25.Cicchetti D, Rogosch F A. (2001) Diverse patterns of neuroendocrine activity in maltreated children. Development and psychopathology. 13(3), 677-693.
- 26.Bruce J, Fisher P A, Pears K C, Levine S. (2009) Morning cortisol levels in preschool-aged foster children: Differential effects of maltreatment type. Doi: 10.1002/dev.20333. Developmental Psychobiology. 51(1),14–23
- 27.Hart J, Gunnar M, Cicchetti D. (1995) Salivary cortisol in maltreated children: Evidence of relations between neuroendocrine activity and social competence. Doi: 10.1017/S0954579400006313. Development and Psychopathology 7(1), 11-26.
- 28.Fisher P A, Kim H K, Bruce J, Pears K C. (2011) Cumulative effects of prenatal substance exposure and early adversity on foster children’s HPA-axis reactivity during a psychosocial stressor. , International Journal of Behavioral Development 36, 29-35.
- 29.Fries E, Hesse J, Hellhammer J, Hellhammer D H. (2005) A new view on hypocortisolism. Doi: 10.1016/j.psyneuen.2005.04.006. Psycho neuroendocrinology 30(10), 1010-1016.
- 30.Kroupina M, Gunnar M R, Johnson D E. Institute of Child Development. University of Minnesota (1997) Report on Salivary Cortisol Levels in a Russian Baby Home. , Minneapolis, MN
- 31.Bruce J, Fisher P A, Pears K C, Levine S. (2009) Morning cortisol Levels in preschool-aged foster children: differential effects of maltreatment type. , Dev Psychobiol 51(1), 14-23.
- 32.Trickett P K, Noll J G, Susman E J, Shenk C E, Putnam F W. (2010) Attenuation of cortisol across development for victims of sexual abuse. Doi: 10.1017/S0954579409990332. Development and psychopathology 22(1), 165-175.
- 33.Brand S R, Brennan P A, Newport D J, Smith A K, Weiss T. (2010) The impact of maternal childhood abuse on maternal and infant HPA axis function in the postpartum period. Doi: 10.1016/j.psyneuen.2009.10.009. Psychoneuroendocrinology. 35(5),686-693
- 34.Fisher D, Serbin L A, Stack D M, Ruttle P L, Ledingham J E. (2007) Intergenerational predictors of diurnal cortisol secretion in early childhood. Doi: 10.1002/icd.474. Infant and Child Development: An International Journal of Research and Practice 16(2), 151-170.
- 35.Yehuda R, Engel S M, Brand S R, Seckl J, Marcus S M. (2005) Transgenerational effects of posttraumatic stress disorder in babies of mothers exposed to the world trade center attacks during pregnancy. , The Journal of Clinical Endocrinology & Metabolism.doi-10.1210/jc.2005-0550 90(7), 4115-4118.
- 36.Dozier M, Bernard K. (2017) Attachment and Biobehavioral Catch‐up: Addressing the needs of infants and toddlers exposed to inadequate or problematic caregiving. Doi: 10.1016/j.copsyc.2017.03.003. Current Opinion in Psychology 15, 111-117.
- 37.. National Scientific Council on the Developing Child. (2005/2014). Excessive Stress Disrupts the Architecture of the Developing Brain: Working Paper 3. Updated Edition.http://www.developingchild.harvard.edu .
- 38.National Scientific Council. (2014) Excessive stress disrupts the development of brain architecture". , Journal of Children's Services.Vol.doi.org/10.1108/JCS-01-2014-0006 9(2), 143-153.
- 39.Spangler G, Schieche M, Ilg U, Maier U, Ackermann C. (1994) Maternal sensitivity as an external organizer for biobehavioral regulation in infancy. Doi: 10.1002/dev.420270702. Developmental Psychobiology 27(7), 425-437.
- 40.Hostinar C E, Gunnar M R. (2013) Future directions in the study of social relationships as regulators of the HPA axis across development. Doi: 10.1080/15374416.2013.804387. , Journal of Clinical Child and Adolescent Psychology 42(4), 564-575.
- 41.Laurent H K, Harold G T, Leve L, Shelton K H, van Goozen SH. (2016) Understanding the unfolding of stress regulation in infants. Doi: 10.1017/S0954579416000171. Development and Psychopathology. 28(4pt2): 1431-1440.
- 42.Levine S. (2005) Developmental determinants of sensitivity and resistance to stress. Doi: 10.1016/j.psyneuen.2005.03.013. Psychoneuroendocrinology. 30(10),939–946
- 43.Gunnar M R, Larson M C, Hertsgaard L, Harris M, Brodersen L. (1992) The stressfulness of separation among 9-month-old infants: Effects of social context variables and infant temperament. Doi: 10.1111/j.1467-8624.1992.tb01627.x. Child Development. 63(2), 290-303.
- 44.Enlow M B, King L, Schreier H M, Howard J M, Rosenfield D. (2014) Maternal sensitivity and infant autonomic and endocrine stress responses. Doi: 10.1016/j.earlhumdev.2014.04.007. Early human development. 90(7), 377-385.
- 45.Haley D W, Stansbury K. (2003) Infant stress and parent responsiveness: Regulation of physiology and behavior during still-face and reunion. doi: 10.1111/1467-8624.00621. Child development. 74(5), 1534-1546.
- 46.Nachmias M, Gunnar M, Mangelsdorf S, Parritz R, Buss K. (1996) Behavioral inhibition and stress reactivity: moderating role of attachment security. doi: 10.1111/j.1467-8624.1996.tb01748.x. Child development. 67(2), 508-522.
- 47.L A Sroufe. (1996) Emotional development: The organization of emotional life in the early years.New York:CambridgeUniversityPress.
- 48.Ahnert L, Gunnar M, Lamb M, Barthel M. (2004) Transition to child care: associations with infant-mother attachment, infant negative emotion and cortisol elevations. Doi: 10.1111/j.1467-8624.2004.00698.x. Child development. 75(3), 639-650.
- 49.Cicchetti D, Rogosch F A, Toth S L, Sturge-Apple M L. (2011) Normalizing the development of cortisol regulation in maltreated infants through preventive interventions. Doi: 10.1017/S0954579411000307. Development and Psychopathology 23(3), 789-800.
- 50.H K Laurent, G T Harold, Leve L, K H Shelton, Goozen S H Van. (2016) Understanding the unfolding of stress regulation in infants. Development and psychopathology. 28(4), 1431-1440.
- 51.Martinez-Torteya C, Dayton C J, Beeghly M, Seng J S, McGinnis E.. Maternal Parenting predicts Infant Biobehavioral Regulation among Women with a History of Childhood Maltreatment. Doi: 10.1017/S0954579414000017. Development and psychopathology 26(2), 379-392.
- 52.Bader K, Hänny C, Schäfer V, Neuckel A, Kuhl C. (2009) . Childhood trauma questionnaire–psychometrische Eigenschaften einer deutschsprachigen Version. Doi: 10.1026/1616-3443.38.4.223. Zeitschrift für Klinische Psychologie und Psychotherapie 38(4), 223-230.
- 53.Bernstein D P, Fink L. (1998) Childhood Trauma Questionnaire. A retrospective self-report. Manual. San Antonio, TX:The Psycho- logical Corporation,Harcourt Brace&Company.
- 54.Ainsworth M D S, M C Blehar, Waters E, Wall S. (1978) Patterns of attachment. A psychological study of the strange situation. Hilsdale. NJ: Lawrence Erlbaurn.
- 55.Bronfman E, Madigan S, Lyons-Ruth K.Atypical Maternal Behavior Instrument for Assessment and Classification (AMBIANCE). Manual for Coding Disrupted Affective Communication,Cambridge,Massachusetts:Department of Psychiatry, Harvard Medical School;1992–2009. Unpublished manual
- 56.Main M, Hesse E. (1990) Parents’ unresolved traumatic experiences are related to infant disorganized attachment status: Is frightened and/or frightening parental behavior the linking mechanism? In. Cummings (Eds.), Attachment in the Preschool Years: Theory, Research, and Intervention (pp. 161 – 182) , Chicago: .
- 57.Lyons-Ruth K, Bronfman E, J Parsons E Vondra, Barnett D. (1999) editors. Atypical maternal behavior and disorganized infant attachment strategies: Frightened, frightening, and atypical maternal behavior and disorganized infant attachment strategies. (Serial No. 258).Atypical patterns of infant attachment: Theory, research, and current directions. Monographs of the Society for Research in Child Development 64(3).
- 58.Kelly K, Slade A, Grienenberger J F. (2005) Maternal reflective functioning, mother–infant affective communication, and infant attachment: Exploring the link between mental states and observed caregiving behavior in the intergenerational transmission of attachment. Doi: 10.1080/14616730500245963. Attachment & human development 7(3), 299-311.
- 59.Perry N B, Mackler J S, Calkins S D, Keane S P. (2014) A transactional analysis of the relation between maternal sensitivity and child vagal regulation. Doi: 10.1037/a0033819. Developmental Psychology 50(3), 784-793.
- 60.Gianino A A, Tronick E Z. (1988) The mutual regulation model: The infant’s self and interactive regulation and coping and defensive capacities. Stress and coping across development. Hillsdale, NJ England: Lawrence Erlbaum Associates, Inc In: Field, TM.; McCabe, PM.; Schneiderman, N.; Field, TM.; McCabe, PM.; Schneiderman, N., editors 47-68.
- 61.DiCorcia J A, Tronick E D. (2011) Quotidian resilience: Exploring mechanisms that drive resilience from a perspective of everyday stress and coping. Doi: 10.1016/j.neubiorev.2011.04.008. Neuroscience & Biobehavioral Reviews 35(7), 1593-1602.
- 62.S W Porges. (1995) Cardiac vagal tone: a physiological index of stress. , Neuroscience & Biobehavioral Reviews.https://doi.org/10.1016/0149-7634(94)00066-A 19(2), 225-233.
- 63.S W Porges, J A Doussard-Roosevelt, A K Maiti. (1994) Vagal Tone and the Physiological Regulation of Emotion. Doi: 10.1111/j.1540-5834.1994.tb01283.x. Monographs of the society for research in child development. 59(23), 167-186.
- 64.Pillhofer M, Spangler G, Bovenschen I, Künster A K, Gabler S. (2015) Pilot study of a program delivered within the regular servicesystem in Germany: Effect of a short-term attachment-basedintervention on maternal sensitivity in mothers at risk for child abuse and neglect. Doi: 10.1016/j.chiabu.2014.07.007. Child Abuse & Neglect 42, 163-173.
Cited by (7)
This article has been cited by 7 scholarly works according to:
Citing Articles:
Brain Sciences (2024) OpenAlex
Franziska Koehler-Dauner, Lena Peter, Eva Roder, Jörg M Fegert, Ute Ziegenhain et al. - Brain Science (2024) Semantic Scholar
Family Relations (2023) OpenAlex
Family Relations (2023) Crossref
F. Köhler-Dauner, U. Ziegenhain, Miriam Kipping, I. Mayer, J. Fegert et al. - Family Relations (2023) Semantic Scholar
Frontiers in Human Neuroscience (2022) OpenAlex
Frontiers in Human Neuroscience (2022) Crossref
A. Buchheim, U. Ziegenhain, H. Kindler, C. Waller, H. Gündel et al. - Frontiers in Human Neuroscience (2022) Semantic Scholar
